Hexavalent chromium exposure5/1/2023  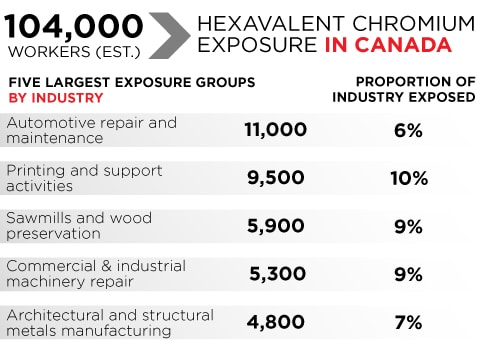
Animal trials show that glutathion plays an important role in Cr(VI) reduction in erythrocytes, also showing certain reduction activity in the lungs ( Suzuki and Fukuda, 1990).īreathing contaminated workplace air is the main source in occupational setting. The main protection mechanism against Cr(VI) activity in the lungs and the stomach is the extracellular reduction of Cr(VI) to Cr(III) by a NADPH-dependent mechanism involving ascorbate ( De Flora et al., 2000). The details of Cr(VI) toxic activity assumed that genotoxicity, including a wide variety of effects such as DNA damage, gene mutation, sister chromatid exchange, chromosomal aberrations, cell transformation, and dominant lethal mutations, may be due to the reduced forms of intracellular origin, formed by the reduction of Cr(VI) to Cr(III) ( Stearns et al., 1995). After absorption, mainly via inhalation for workers and/or via ingestion for the general population, Cr(VI) readily penetrates cell membranes. The toxicity of Cr(VI) in humans has been reviewed extensively ( ATSDR, 2012 Costa and Klein, 2006 U.S. 1B or 1A).Īlso the dermal exposure to Cr(VI) compounds can cause skin irritation, ulceration, sensitization, and allergic contact dermatitis ( NIOSH, 2002). In the EU Classification, Labelling and Packaging (CLP) Regulation (EC) No 1272/2008, Cr(VI) are classified as genotoxic (Muta. 
In the European Union (EU), the estimated number of Cr(VI)-exposed workers in 2012 was ~786,000, with the largest numbers exposed to welding ( IARC, 2012). Hexavalent chromium was classified by the International Agency for Research on Cancer (IARC) as a human carcinogen (Group 1) associated with increased lung cancer risk among workers in certain industries and also cancer of the nose and nasal sinuses. Dermal exposure through leather articles and cosmetics, and oral exposure of children through toys have been reported. Other sources of exposure to Cr(VI) need to be considered for general population, including the release of Cr, with Cr(VI) as the predominant species, from orthopedic implants made from stainless steel and cobalt-chromium alloys. For more detailed information on chromium concentrations in these environmental compartments, please consult the HBM4EU Scoping document on cadmium and chromium VI. Mobilisation of Cr occurs mainly through air, water and soil. A report on a critical raw material profile by the European Commision in 2014 revealed that the forecast average annual demand for Cr growth of 3%-4.5% per year ( EC Report, 2014, Report on Critical raw materials for the EU). Within the EU, Finland was the main producer of Cr in 2006, producing over 99% of the total EU Cr production (219,500 tonnes). The main sources for EU imports in 2006 were South Africa (approximately 80%). Due to a lack of internal supply and to demand from the steel industry, the EU has been an importer of Cr ores. Major uses of Cr(VI) compounds include metal plating, manufacture of pigments and dyes, corrosion inhibitors, chemical synthesis, refractory production, leather tanning, and wood preservation ( Blade et al., 2007). Most of Cr(VI) compounds are man-made (products or by-products) and human-caused Cr(VI) contamination is a result of large industrial emissions (mainly from metallurgical, chemical, and refractory brick industries). 
The occurrence of Cr(VI) is rare naturally. Hexavalent form – Cr(VI) – is more toxic than trivalent form – Cr(III) for its high oxidizing potential – and easily penetrates biological membranes. The +3 and +6 forms are the most important as the +2, +4, and +5 forms are unstable and are rapidly converted to +3, which in turn is oxidized to +6 ( Towill et al. Chromium can exist in oxidation states ranging from -2 to +6, but is most frequently found in the environment in the trivalent (+3) and hexavalent (+6) oxidation states. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
